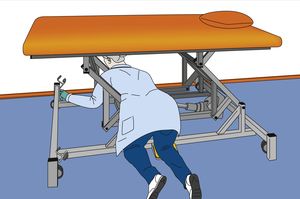
In December 2020, the supreme regional authorities in Germany responsible for medical devices and the German Federal Institute for Drugs and Medical Devices (BfArM) published a new document containing information on the safety of treatment tables and corresponding requirements. One of the requirements upon manufacturers that is addressed in the document is that they observe the updated BfArM recommendation for treatment tables with power adjustment to be designed such that the tables are not able to trap persons and thereby cause serious injury. A guide for operators is also included, providing information on the purchase, operation and use of treatment tables with electrical height adjustment.
The reason for production of the new document is the risk of persons being trapped beneath a treatment table with electrical height adjustment, resulting in injury and possibly even death, see also KANBrief 4/20.
The document is available for download (in German), for example on the website of the Münster district government (pdf, non-accessible)
 © BG BAU/Steindesign Werbeagentur
© BG BAU/Steindesign Werbeagentur